Durvalumab Investigator Brochure
Durvalumab Investigator Brochure - On may 1, 2017, the u.s. For more information on immunotherapy medications, click here. Durvalumab is an immunotherapy medication. Alongside the updated protocol, we are also introducing some new and updated supporting documents. B2 durvalumab + investigator's choice of chemotherapy + danvatirsen Nccn guidelines · ordering · hcp & patient materials · kol videos Please contact the rampart team. Please contact the rampart team. This was followed by durvalumab or placebo every 4 weeks for up to 12 cycles after surgery. Several payment sources exist for cancer drugs in ontario, depending. This was followed by durvalumab or placebo every 4 weeks for up to 12 cycles after surgery. B2 durvalumab + investigator's choice of chemotherapy + danvatirsen Imfinzi may be used when: Fda approvedprescribing informationcontinuous dosingsafety information As the durvalumab and tremelimumab investigator brochures contain confidential information, they are kept within the member's area of the website. Durvalumab is an immunotherapy medication. Alongside the updated protocol, we are also introducing some new and updated supporting documents. The durvalumab investigator brochure (ib) has recently been updated by astrazeneca and there are new expected toxicities listed for both durvalumab monotherapy and the combination with. The primary endpoint of the trial was event free survival (efs). Several payment sources exist for cancer drugs in ontario, depending. Durvalumab is an immunotherapy medication. Please contact the rampart team. The durvalumab investigator brochure (ib) has recently been updated by astrazeneca and there are new expected toxicities listed for both durvalumab monotherapy and the combination with. As the durvalumab and tremelimumab investigator brochures contain confidential information, they are kept within the member's area of the website. Alongside the updated protocol,. Imfinzi may be used when: Durvalumab is an immunotherapy medication. For more information on immunotherapy medications, click here. Alongside the updated protocol, we are also introducing some new and updated supporting documents. Imfinzi™ (durvalumab) is a prescription medicine used to treat a type of cancer in the bladder and urinary tract called urothelial carcinoma. Fda approvedprescribing informationcontinuous dosingsafety information The durvalumab investigator brochure (ib) has recently been updated by astrazeneca and there are new expected toxicities listed for both durvalumab monotherapy and the combination with. Please contact the rampart team. The durvalumab investigator brochure (ib) has recently been updated by. Alongside the updated protocol, we are also introducing some new and updated supporting documents. The durvalumab investigator brochure (ib) has recently been updated by. Please contact the rampart team. Study protocol has updated to align and be consistent with the broader durvalumab programme, including the investigator brochure and the clinical study protocol format of more recent. Fda approvedprescribing informationcontinuous dosingsafety information Alongside the updated protocol, we are also introducing some new and updated supporting. The durvalumab investigator brochure (ib) has recently been updated by. Imfinzi™ (durvalumab) is a prescription medicine used to treat a type of cancer in the bladder and urinary tract called urothelial carcinoma. B2 durvalumab + investigator's choice of chemotherapy + danvatirsen As the durvalumab and tremelimumab investigator brochures contain confidential information, they are kept within the member's area of the. Please contact the rampart team. On may 1, 2017, the u.s. Food and drug administration granted accelerated approval to durvalumab (imfinzi, astrazeneca uk limited) for the treatment of patients with locally. Imfinzi™ (durvalumab) is a prescription medicine used to treat a type of cancer in the bladder and urinary tract called urothelial carcinoma. Please contact the rampart team. Nccn guidelines · ordering · hcp & patient materials · kol videos For more information on immunotherapy medications, click here. On may 1, 2017, the u.s. As the durvalumab and tremelimumab investigator brochures contain confidential information, they are kept within the member's area of the website. The primary endpoint of the trial was event free survival (efs). This was followed by durvalumab or placebo every 4 weeks for up to 12 cycles after surgery. Several payment sources exist for cancer drugs in ontario, depending. Nccn guidelines · ordering · hcp & patient materials · kol videos The primary endpoint of the trial was event free survival (efs). As the durvalumab and tremelimumab investigator brochures contain confidential information,. Food and drug administration granted accelerated approval to durvalumab (imfinzi, astrazeneca uk limited) for the treatment of patients with locally. Please contact the rampart team. As the durvalumab and tremelimumab investigator brochures contain confidential information, they are kept within the member's area of the website. Several payment sources exist for cancer drugs in ontario, depending. Imfinzi™ (durvalumab) is a prescription. Astrazeneca’s imfinzi (durvalumab) in combination with gemcitabine and cisplatin as neoadjuvant treatment, followed by imfinzi as adjuvant monotherapy after radical. Please contact the rampart team. This was followed by durvalumab or placebo every 4 weeks for up to 12 cycles after surgery. For more information on immunotherapy medications, click here. The primary endpoint of the trial was event free survival. Alongside the updated protocol, we are also introducing some new and updated supporting documents. As the durvalumab and tremelimumab investigator brochures contain confidential information, they are kept within the member's area of the website. Astrazeneca’s imfinzi (durvalumab) in combination with gemcitabine and cisplatin as neoadjuvant treatment, followed by imfinzi as adjuvant monotherapy after radical. Nccn guidelines · ordering · hcp & patient materials · kol videos Imfinzi™ (durvalumab) is a prescription medicine used to treat a type of cancer in the bladder and urinary tract called urothelial carcinoma. Imfinzi may be used when: On may 1, 2017, the u.s. As the durvalumab and tremelimumab investigator brochures contain confidential information, they are kept within the member's area of the website. For more information on immunotherapy medications, click here. The durvalumab investigator brochure (ib) has recently been updated by astrazeneca and there are new expected toxicities listed for both durvalumab monotherapy and the combination with. Several payment sources exist for cancer drugs in ontario, depending. Please contact the rampart team. Food and drug administration granted accelerated approval to durvalumab (imfinzi, astrazeneca uk limited) for the treatment of patients with locally. Durvalumab is an immunotherapy medication. The durvalumab investigator brochure (ib) has recently been updated by. Please contact the rampart team.(PDF) Firstline durvalumab and tremelimumab with chemotherapy in RAS
FDA a aprobat durvalumab, prima imunoterapie pentru carcinomul pulmonar
Durvalumab Plus Chemotherapy for Advanced Biliary Tract Cancer
Thuốc Durvalumab Công dụng và những điều cần lưu ý
(PDF) Concurrent durvalumab and radiation therapy (DUART) followed by
Figure 1 from Evaluating the Therapeutic Potential of Durvalumab in
DatoDXd + Durvalumab + Carboplatin for Advanced NonSmall Cell Lung
Figure 1 from Durvalumab in NSCLC latest evidence and clinical
(PDF) Durvalumab Plus Carboplatin/Paclitaxel Followed by Maintenance
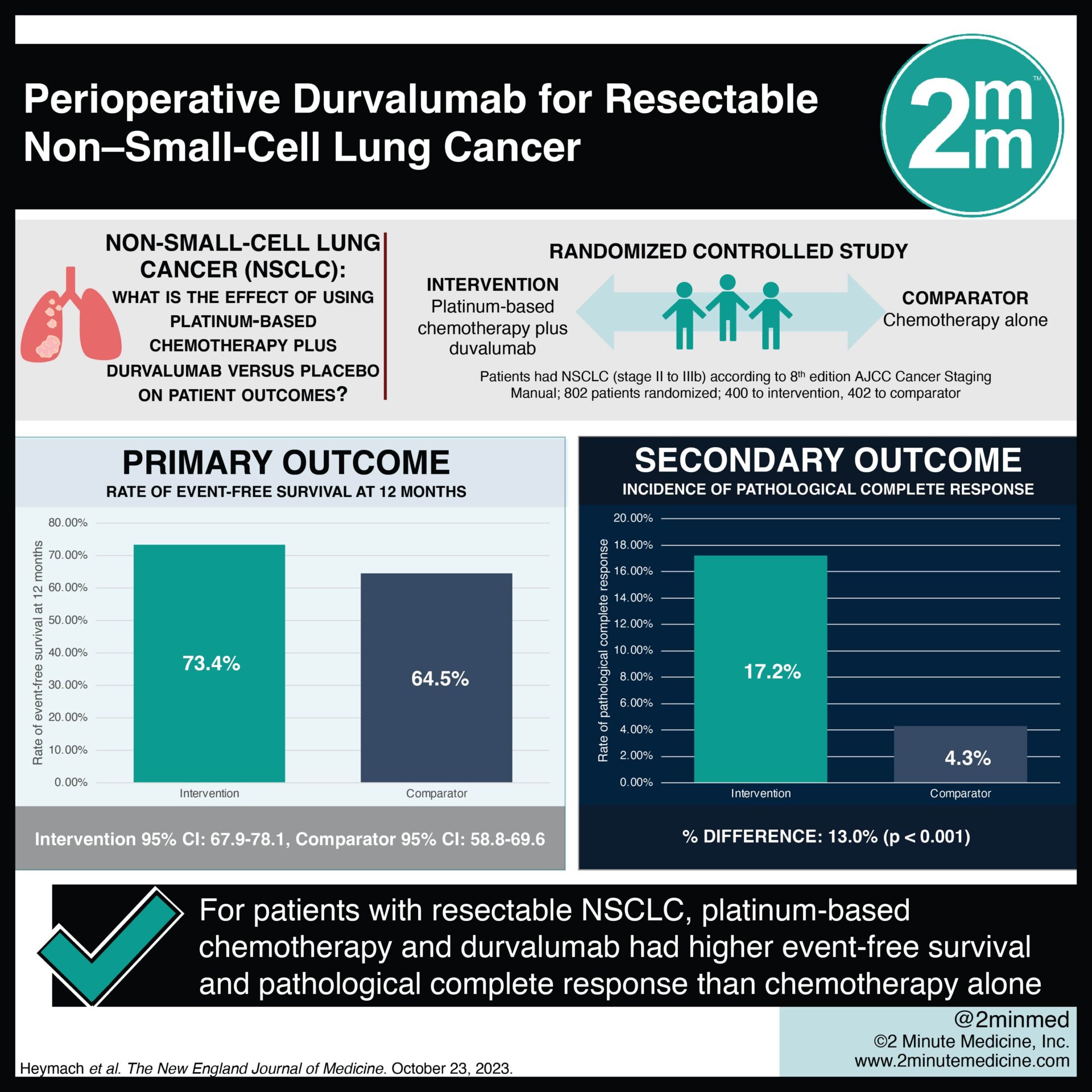
VisualAbstract Perioperative Durvalumab for Resectable NonSmallCell
This Was Followed By Durvalumab Or Placebo Every 4 Weeks For Up To 12 Cycles After Surgery.
Fda Approvedprescribing Informationcontinuous Dosingsafety Information
Study Protocol Has Updated To Align And Be Consistent With The Broader Durvalumab Programme, Including The Investigator Brochure And The Clinical Study Protocol Format Of More Recent.
B2 Durvalumab + Investigator's Choice Of Chemotherapy + Danvatirsen
Related Post:


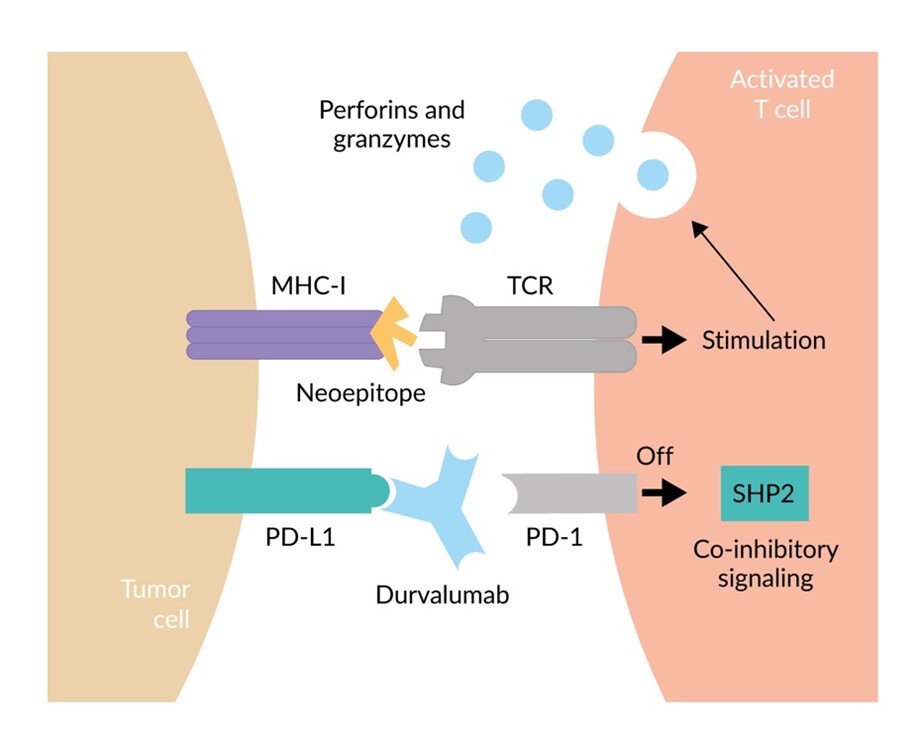



:%0A%0ADato-DXd + Durvalumab + Carboplatin for Advanced Non-Small Cell Lung Cancer.png?md=1)